Techniques and Methods
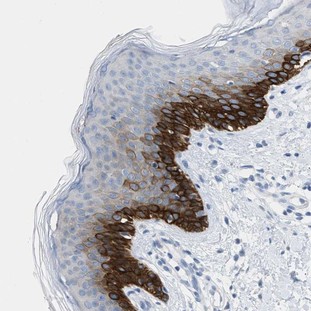
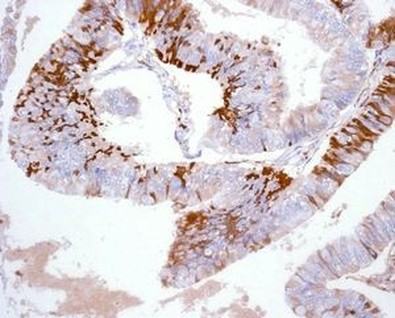
IMMUNOHISTOCHEMISTRY
This technique is
fundamental for the better understanding of the Angiotensin-Renin system and
kinins. Thanks to this method we can localize all the necessary components for
the synthesis of AGII, bradykinins and even of the respective receptors. Through
this technique the local synthesis of AGII in the hearth
and brain has been discovered.
Immunohistochemistry consists of the use of antibodies that connect to specific epitopes of the antigens we want to study. They are afterwards put to react to peroxidases or other enzymes that allow the dyeing of the antibodies. We can now localize the action site of the antigen. Antibodies can also be marked with fluorescent molecules.
It´s also important to notice the difference between immunohistochemistry and immunocytochemistry. Both the procedures use the antibody/antigen relation, but the former is used in tissue fixated by physical or chemical methods. The immnocytochemistry is used primarily in smears.
To produce the monoclonal antibodies, desired for their great specificity, we extract from a infected rat, lymphocytes B that can produce antibodies. Because lymphocytes have a very short life outside of our organism, they are fused with myelomas (plasmocytes with neoplastic activity) They can, because of that, duplicate very quikly.
The hybrid cells born out of this fusion are denominated hybridomas and can produce a enormous quantity of antibodies.
The final step consists in separating the hybridomas so we can obtained a group of cells that can produced monoclonal antibodies, meaning, antibodies that come from clones of a unique lymphocyte and only react to a specific epitope.
Another method to increase the precision of the immunohistochemistry process is using a method involving two steps. This means that we use a antiboidie to react with the antigen and then we use another that will connect to the initial antibody.
Immunohistochemistry consists of the use of antibodies that connect to specific epitopes of the antigens we want to study. They are afterwards put to react to peroxidases or other enzymes that allow the dyeing of the antibodies. We can now localize the action site of the antigen. Antibodies can also be marked with fluorescent molecules.
It´s also important to notice the difference between immunohistochemistry and immunocytochemistry. Both the procedures use the antibody/antigen relation, but the former is used in tissue fixated by physical or chemical methods. The immnocytochemistry is used primarily in smears.
To produce the monoclonal antibodies, desired for their great specificity, we extract from a infected rat, lymphocytes B that can produce antibodies. Because lymphocytes have a very short life outside of our organism, they are fused with myelomas (plasmocytes with neoplastic activity) They can, because of that, duplicate very quikly.
The hybrid cells born out of this fusion are denominated hybridomas and can produce a enormous quantity of antibodies.
The final step consists in separating the hybridomas so we can obtained a group of cells that can produced monoclonal antibodies, meaning, antibodies that come from clones of a unique lymphocyte and only react to a specific epitope.
Another method to increase the precision of the immunohistochemistry process is using a method involving two steps. This means that we use a antiboidie to react with the antigen and then we use another that will connect to the initial antibody.
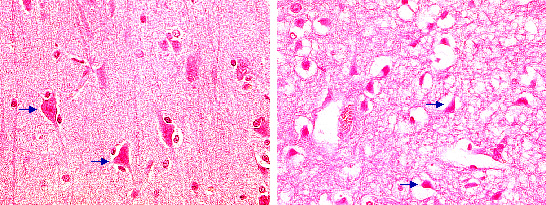
Fig. 24: Hematoxylin-eosin (HE) stained brain sections: Left; typical staining pattern of healthy neurons with reddish appearance of the cell body and processes. Right: Neurons following experimental stroke with irreversible cell damage. Note shrinkage of the nucleus and vacuolization of the cytoplasm, collagen fibers and other protein rich structures