Procedures
1st Stage
 Fig. 20: Measure of blood pressure.
Fig. 20: Measure of blood pressure.
During the first
stage, in collaboration with competent authorities in the proper capture and
treatment of the species of bat lesser horseshoe bat (Rhinolophus hipposideros), we intend to study
their morphophysiology and hormonal profile through a non-invasive method.
To achieve this purpose, we begin by capturing wild specimens with the help of experts in the field, in order to ensure their welfare and proper handling. Next we intend, through non-invasive techniques, to monitor their intracranial blood pressure and keep track of the hormonal composition of their blood through adequate tests.
In order to accurately measure the intravenous pressure our method of choice would be a subdural screw. This method allows the measurement of blood pressure in real time. In the procedure a hollow screw is inserted through a hole drilled in the skull and placed in “dura mater”. This way the sensor can record from inside the subdural space, accurately measuring the pressure. In order to cause the least amount of damage and stress to the animal, the whole procedure will be made by qualified veterinarians. Regarding the blood tests, to identify the hormones involved in the regulation of blood pressure, we will use a blood chemistry test in which we will isolate the blood plasma (where the chemicals in the blood are dissolved) by centrifugation. In the next step we can use a spectrophotometer to obtain the photometric spectrum of the plasma. For this we will use plasma from bats with little rest time as well as from bats after short or non-existent resting periods (and thus has been hanging upside down for a long span of time).It is therefore possible to compare the spectra, analyze the differences and then pinpoint the required hormones. Now that we have identified them we can produce specific antibodies or use chemicals with affinity to the hormones and see their effect when applied to lab rats.
To achieve this purpose, we begin by capturing wild specimens with the help of experts in the field, in order to ensure their welfare and proper handling. Next we intend, through non-invasive techniques, to monitor their intracranial blood pressure and keep track of the hormonal composition of their blood through adequate tests.
In order to accurately measure the intravenous pressure our method of choice would be a subdural screw. This method allows the measurement of blood pressure in real time. In the procedure a hollow screw is inserted through a hole drilled in the skull and placed in “dura mater”. This way the sensor can record from inside the subdural space, accurately measuring the pressure. In order to cause the least amount of damage and stress to the animal, the whole procedure will be made by qualified veterinarians. Regarding the blood tests, to identify the hormones involved in the regulation of blood pressure, we will use a blood chemistry test in which we will isolate the blood plasma (where the chemicals in the blood are dissolved) by centrifugation. In the next step we can use a spectrophotometer to obtain the photometric spectrum of the plasma. For this we will use plasma from bats with little rest time as well as from bats after short or non-existent resting periods (and thus has been hanging upside down for a long span of time).It is therefore possible to compare the spectra, analyze the differences and then pinpoint the required hormones. Now that we have identified them we can produce specific antibodies or use chemicals with affinity to the hormones and see their effect when applied to lab rats.
With
this procedures it will be of the uttermost importance to methodically track hormonal fluctuations by following
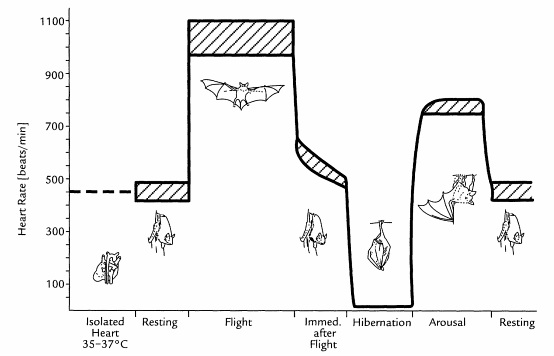
a fitting schedule. Our schedule is based on a study (Neuweiller, 2000)
which allowed the construction of the graphic bellow.
In
order to track
hormonal fluctuations, the blood composition and pressure measurements will be
performed in bats during rest (at different times, to accurately measure the
fluctuations) and in bats immediately after flight and deferent stages of
hibernation.
The blood analyses will provide us with information about the blood composition of bats. This area of study remains largely unexplored (Neuweiller, 2000), having extremely importance, not only for our study, but also to understand the physiology of the circulatory system of bats and the physiology of flight. At this point, our team will establish a partnership with biologists interested in this area, hoping to promote and develop studies in this field.
After the analyses, we can expect to identify and isolate two hormones (denominated H1 and H2, on a non-compromised way) related do the circulatory system of this animal, responsible for the relaxation and constriction of the blood vessels.
With this information at hand we can proceed to the next stage of the experiment.
The blood analyses will provide us with information about the blood composition of bats. This area of study remains largely unexplored (Neuweiller, 2000), having extremely importance, not only for our study, but also to understand the physiology of the circulatory system of bats and the physiology of flight. At this point, our team will establish a partnership with biologists interested in this area, hoping to promote and develop studies in this field.
After the analyses, we can expect to identify and isolate two hormones (denominated H1 and H2, on a non-compromised way) related do the circulatory system of this animal, responsible for the relaxation and constriction of the blood vessels.
With this information at hand we can proceed to the next stage of the experiment.
2nd Stage
 Fig. 21: Example of an industrial centrifuge.
Fig. 21: Example of an industrial centrifuge.
The second stage of our experiment consists of two distinct steps. In the first one, and based on previous experiments with the human clinical trials (about which we provide further details in the following paragraph), we will study the effects of these already identified hormones in the lab rats, subjected to our particular experimental approach. This will allow us to establish a comparative reference, providing us with a way to verify the effectiveness of the previously identified hormones, which will be the subject of the second stage.
Hormone therapies are widely used in the treatment of various pathologies. An example of an hormone therapy, deeply connected to our project, is the use of estrogens as an experimental therapy to treat strokes (Ouyang, et. al. 2006); however, there were reported some problems on the clinical trials of this project, one of them was the “increase of systemic angiotensin II, a potent vasoconstrictor” (Prime, et. al. 2007). Immediately, other studies suggested that the angiotensin-renin system could be the alternative therapy for this diseases (McCarthy, et. al. 2013 and Saavedra, 2005), however, because they are recent studies, there is a lot more to explore, and, with our experimental method, we want to confirm these hypotheses in order to ensure the viability of this therapy.
Having defined distinct steps for this stage of the experiment, we can now provide a detailed walkthrough of each of these two phases. Given that both will consist of the same methodology, the only difference being the type of hormones applied, we can start by outlining the experimental method used throughout the experiment.
In order to simulate constant pressure with great control over the extend as well as the intensity of the force applied to the specimen, we opted to use an adapted centrifuge instead of the classic pressure chamber. By locking a rat’s body tightly in each compartment of the centrifuge with the head directed the furthest away from the rotational center of the machine, the blood will be forcefully rushed towards the brain and so exert control pressure in this area. This enables us to redirect the blood flow to specific parts of the rat’s anatomy (in this case, the cranium), as opposed to the non-localized full body effect that a pressure chamber would create, thus having the advantage of providing a more restricted area of effect. Knowing the average mass of the blood present in the rat, we can calculate the force applied per unit of area and therefore the pressure (Pressure=Force/Area) for a given velocity using the formula:
Hormone therapies are widely used in the treatment of various pathologies. An example of an hormone therapy, deeply connected to our project, is the use of estrogens as an experimental therapy to treat strokes (Ouyang, et. al. 2006); however, there were reported some problems on the clinical trials of this project, one of them was the “increase of systemic angiotensin II, a potent vasoconstrictor” (Prime, et. al. 2007). Immediately, other studies suggested that the angiotensin-renin system could be the alternative therapy for this diseases (McCarthy, et. al. 2013 and Saavedra, 2005), however, because they are recent studies, there is a lot more to explore, and, with our experimental method, we want to confirm these hypotheses in order to ensure the viability of this therapy.
Having defined distinct steps for this stage of the experiment, we can now provide a detailed walkthrough of each of these two phases. Given that both will consist of the same methodology, the only difference being the type of hormones applied, we can start by outlining the experimental method used throughout the experiment.
In order to simulate constant pressure with great control over the extend as well as the intensity of the force applied to the specimen, we opted to use an adapted centrifuge instead of the classic pressure chamber. By locking a rat’s body tightly in each compartment of the centrifuge with the head directed the furthest away from the rotational center of the machine, the blood will be forcefully rushed towards the brain and so exert control pressure in this area. This enables us to redirect the blood flow to specific parts of the rat’s anatomy (in this case, the cranium), as opposed to the non-localized full body effect that a pressure chamber would create, thus having the advantage of providing a more restricted area of effect. Knowing the average mass of the blood present in the rat, we can calculate the force applied per unit of area and therefore the pressure (Pressure=Force/Area) for a given velocity using the formula:
It is thus possible to make fine adjustment to the centrifuge’s speed in order to oscillate the blood pressure as required and therefore feasible to test a number of variables for a given hormone’s concentration. By keeping track of the rats’ condition throughout the experiment as well as their survival rate, we will be able to conclude which hormone is the most effective in reducing the intravascular pressure.
Since the not only the intensity of the pressure but also its duration can have a great impact on the subject’s brain, it will be necessary to account for a number of different experimental conditions.
Since the not only the intensity of the pressure but also its duration can have a great impact on the subject’s brain, it will be necessary to account for a number of different experimental conditions.
-
Step A
In this step, as already stated above, our efforts
will be focused on testing the survival rate of the rat subjected to high
intracranial pressures after the administration of previously documented human
hormones (so the results may be fairly predictable), which will serve as a
reference for the newfound hormone’s effectiveness. It’s also essential to have
an exact control group for the same reason. It’s also important to see how the
brain adapts to both very high pressures in short bursts and moderately high
pressure sustained over a longer time span, so the number of experimental
groups will be doubled accordingly.
|
Experimental groups (very high pressure/short
exposure and high pressure/ long exposure):
1. Control without injected hormones nor centrifugation; 2. Control without injected hormone and with centrifugation; Not subjected to centrifugation: 3. Injected with angiotensin; 4. Injected with kinins; 5. Injected with both angiotensin and kinins; Subjected to centrifugation: 6. Injected with angiotensin; 7. Injected with kinins; 8. Injected with both angiotensin and kinins |
It’s also important to learn what happens in the total absence of these hormones. “Knock out” subjects can be used to accomplish such task, as follows:
9. “Knock out” control without injected hormones nor centrifugation; 10. “Knock out” control without injected hormone and with centrifugation; Not subjected to centrifugation: 11. “Knock out” injected with angiotensin; 12. “Knock out” injected with kinins; 13. “Knock out” injected with both angiotensin and kinins; Subjected to centrifugation: 14. “Knock out” injected with angiotensin; 15. “Knock out” injected with kinins; 16. “Knock out” injected with both angiotensin and kinins |
- Step B
We can now test the
effectiveness of the new hormones provided by the bats. It’s important to
expose the specimens to the exact same experimental condition as in the
previous step, so as to make sure that the only variable of the study is in
fact the hormone utilized.
|
Experimental groups (very high pressure/short
exposure and high pressure/ long exposure):
1. Control without injected hormones nor centrifugation; 2. Control without injected hormone and with centrifugation; Not subjected to centrifugation: 3. Injected with H1; 4. Injected with H2; 5. Injected with both H1 and H2; Subjected to centrifugation: 6. Injected with H1; 7. Injected with H2; 8. Injected with both H1 and H2 |
It’s also importante to learn what happens in the
total absence of these hormones. “Knock out” subjects can be used to acomplish
such task, as follows:
9. “Knock out” control without injected hormones nor centrifugation; 10. “Knock out” control without injected hormone and with centrifugation; Not subjected to centrifugation: 11. “Knock out” injected with H1; 12. “Knock out” injected with H2; 13. “Knock out” injected with both H1 and H2; Subjected to centrifugation: 14. “Knock out” injected with H1; 15. “Knock out” injected with H2; 16. “Knock out” injected with H1 and H2 |
3rd Stage
Now that we have
performed both steps of the second stage we already know which group performed
better by analyzing the survival/death ration, and how long each specimen was
able to sustain the respective experimental conditions. However it’s now
mandatory to proceed to the observation of the tissue damage in each individual
rat, and to do this histology will be an essential tool. With the proper
guidance provided by the Histologists hired, we can prepare the brain tissues
for microscopic observation with proper fixation and staining. This way, we can
analyses the damage with great precision, as well as identify the main areas of
the brain affected. This information will prove useful for the development of in
situ delivery of hormones and drugs, exponentially improving the treatment’s
effectiveness.